For a lot of chemistry it's very hard to work out from first principles; you just have to learn it. |
|
Results 1 to 5 of 5
-
11-27-2010 03:42 PM #1
Tell me about chemical equations.
How do you find the product of a reaction?
For example: 2Mg+O2 = 2MgO
How come it is 2MgO instead of 2MgO2??
-
11-27-2010 06:19 PM #2Banned


- Join Date
- Aug 2005
- Posts
- 9,984
- Likes
- 3084
-
11-27-2010 06:20 PM #3
If the 2 is subscript, it refers only to the atom is is attached to. If it is in front (as in 2MgO) it refers to the entire molecule. Therefore 2MgO refers to 2 moles of magnesium oxide compound (MgO). 2MgO2 would have 4 total moles of oxygen.
-
11-27-2010 06:51 PM #4
Your question is pretty broad in general (not good for the ask/tell forum), I suggest google...that's where you are most likely to find the answers to the questions you have. Read through this site in regards to balancing chemical equations: Chemical Equations
-
11-27-2010 07:13 PM #5
Chemical engineer student here to the rescue!
Firstly, you are thinking that chemical reactions are like mathematical formulas, they are not! Chemical reactions are a representation of the starting properties and the final properties of the atoms and compounds involved in the reaction, and it must respect the mechanisms that cause the reaction!
The formula itself is just a simplification of what is happening. When you react your Mgs and your Os, they wont simply pair up and stay away from other compounds, they will interact and form a net. These nets will have different amouts of Mgs and Os, but they will respect the most important thing about chemical equations, the ratio between Mgs and Os! When you say that it the compound formed is the MgO, you say that the new compound has a ration of 1:1 of Mgs and Os. The most correct (do not use this, though) way to represent it would be MgxOx, x being the number of atoms in the net. It could be Mg2O2, Mg3O3 or even Mg99999O99999!
Here is a image of the net I'm talking about:
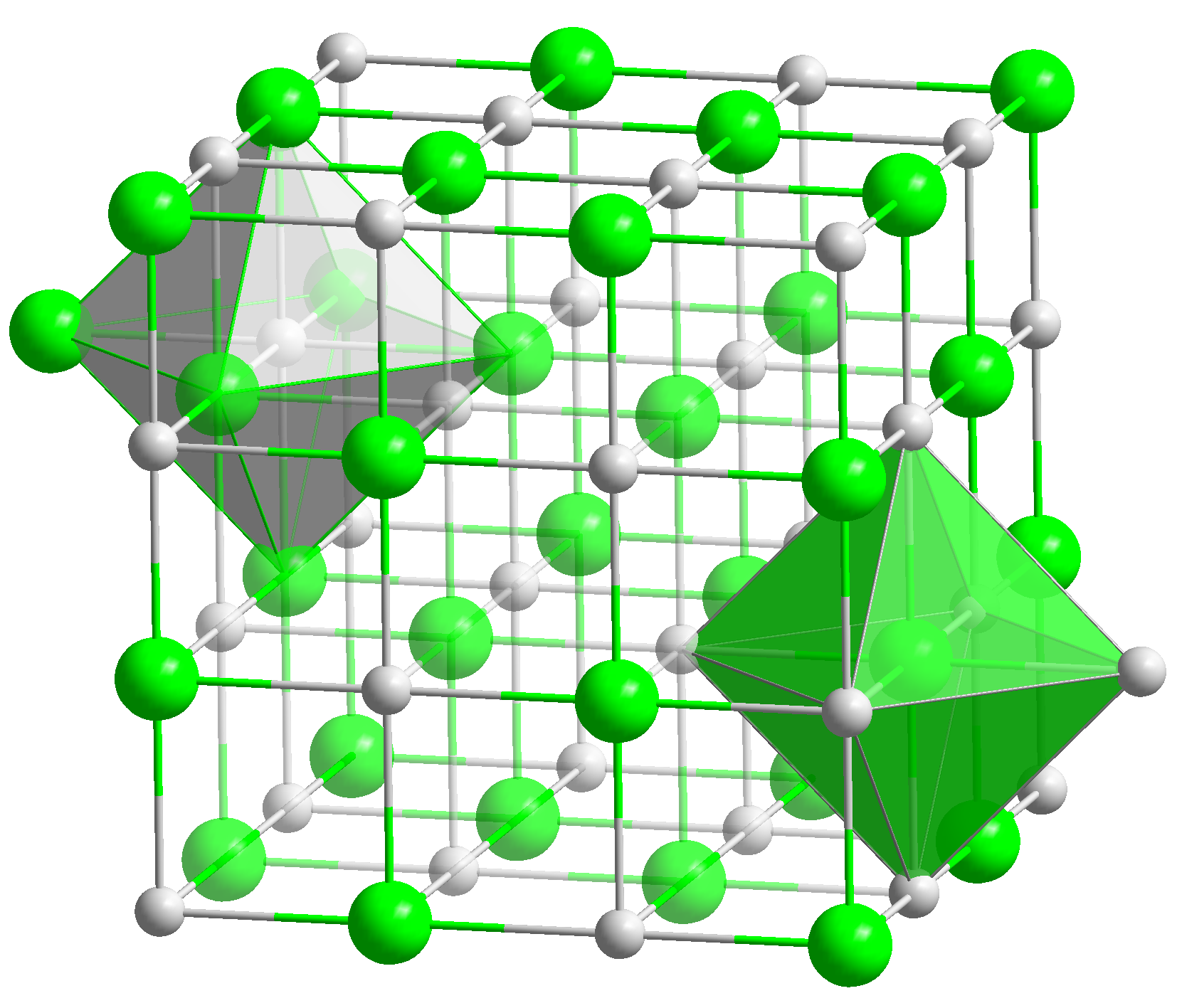
Why can't it be MgO2?
Remember what reactions are? They are when atoms, while trying to reach stability, or the lowest level of energy, exchange electrons. A simple way to understand this is to look at the periodic table. The groups (or columns) are very usefull for understanding who wants to give and who wants to receive electrons. According to the octect rule, all atoms need to fill up their electron shells in order to become stable (this is very simplified if you want to I can explain it better later, but you can use this for most simple reactions). The 18th group is the last group, all atoms there will have 8 electrons in the outmost shell, and that means stability!
The oxigen atom is in the 16th group, that means it has 6 electrons in the outmost shell (the p, or principal, shell)! So it needs to gain 2 electrons, or move 2 groups up to reach the 18th group. So, in a reaction, the oxigen will try to gain 2 electrons.
The magnesium is in the 2nd group. He also wants to reach the stability by acquiring 8 electrons, but, just look at how many electrons he'd have to gain to move all the way to the 18th group of his row! It is much more easy (read 'energetically favorable') to just lose 2 electrons and move down to the previous row! So, by losing 2 electrons, he will have a full p orbital group, and will become stable.
So, the Mg want to give 2 electrons and the O wants to gain 2 electrons! Add the together and you should get a chemical reaction, with the Mg giving O two electrons. They will then attract each other as they will have charges, being Mg positively charged and the O negatively charged, thus forming the net.
MgO2 would not become stable, as it means that a single Mg is giving his 2 electrons to 2 oxigen, so each oxigen would be receiving one electron each, which would mean they woudn't have reached the number of 8 electrons in the outmost shell, thus not becoming stable. The molecule would quickly break down to form a MgO.
Here is the reaction:
ZzzzzZZzzz....

Similar Threads
-
Equations
By [SomeGuy] in forum Science & MathematicsReplies: 71Last Post: 06-11-2009, 04:56 PM -
Equations 2
By ninja9578 in forum Science & MathematicsReplies: 84Last Post: 03-10-2009, 11:56 PM -
The Fire in the Equations
By Xei in forum PhilosophyReplies: 2Last Post: 01-08-2009, 06:33 PM -
Chemical impossibility?
By acheron in forum General Lucid DiscussionReplies: 9Last Post: 09-20-2008, 11:00 PM -
Mathematical Equations
By Sornaensis in forum Senseless BanterReplies: 39Last Post: 03-13-2008, 02:44 AM




 1Likes
1Likes LinkBack URL
LinkBack URL About LinkBacks
About LinkBacks


 Reply With Quote
Reply With Quote



Bookmarks